
"Buckyball"


Buckminsterfullerene
| | This section does not cite any references or sources. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed. (May 2010) |
Buckminsterfullerene (IUPAC name (C60-Ih)[5,6]fullerene) is the smallest fullerene molecule in which no two pentagons share an edge (which can be destabilizing, as in pentalene). It is also the most common in terms of natural occurrence, as it can often be found in soot.
The structure of C60 is a truncated (T = 3) icosahedron, which resembles a soccer ball of the type made of twenty hexagons and twelve pentagons, with a carbon atom at the vertices of each polygon and a bond along each polygon edge.
The van der Waals diameter of a C60 molecule is about 1 nanometer (nm). The nucleus to nucleus diameter of a C60 molecule is about 0.71 nm.
The C60 molecule has two bond lengths. The 6:6 ring bonds (between two hexagons) can be considered "double bonds" and are shorter than the 6:5 bonds (between a hexagon and a pentagon). Its average bond length is 1.4 angstroms.
Silicon buckyballs have been created around metal ions.
Boron buckyball
A new type of buckyball utilizing boron atoms instead of the usual carbon has been predicted and described in 2007. The B80 structure, with each atom forming 5 or 6 bonds, is predicted to be more stable than the C60 buckyball.[13] One reason for this given by the researchers is that the B-80 is actually more like the original geodesic dome structure popularized by Buckminster Fuller which utilizes triangles rather than hexagons. However, this work has been subject to much criticism by quantum chemists[14][15] as it was concluded that the predicted Ih symmetric structure was vibrationally unstable and the resulting cage undergoes a spontaneous symmetry break yielding a puckered cage with rare Th symmetry (symmetry of a volleyball).[14] The number of six atom rings in this molecule is 20 and number of five member rings is 12. There is an additional atom in the center of each six member ring, bonded to each atom surrounding it.
Variations of buckyballs
Another fairly common buckminsterfullerene is C70,[16] but fullerenes with 72, 76, 84 and even up to 100 carbon atoms are commonly obtained.
In mathematical terms, the structure of a fullerene is a trivalent convex polyhedron with pentagonal and hexagonal faces. In graph theory, the term fullerene refers to any 3-regular, planar graph with all faces of size 5 or 6 (including the external face). It follows from Euler's polyhedron formula, |V|-|E|+|F| = 2, (where |V|, |E|, |F| indicate the number of vertices, edges, and faces), that there are exactly 12 pentagons in a fullerene and |V|/2-10 hexagons.
 |  | 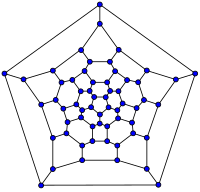 |  |
| 20-fullerene (dodecahedral graph) | 26-fullerene graph | 60-fullerene (truncated icosahedral graph) | 70-fullerene graph |
The smallest fullerene is the dodecahedron – the unique C20. There are no fullerenes with 22 vertices.[17] The number of fullerenes C2n grows with increasing n = 12,13,14..., roughly in proportion to n9 (sequence A007894 in OEIS). For instance, there are 1812 non-isomorphic fullerenes C60. Note that only one form of C60, the buckminsterfullerene alias truncated icosahedron, has no pair of adjacent pentagons (the smallest such fullerene). To further illustrate the growth, there are 214,127,713 non-isomorphic fullerenes C200, 15,655,672 of which have no adjacent pentagons.
Trimetasphere carbon nanomaterials were discovered by researchers at Virginia Tech and licensed exclusively to Luna Innovations. This class of novel molecules comprises 80 carbon atoms (C80) forming a sphere which encloses a complex of three metal atoms and one nitrogen atom. These fullerenes encapsulate metals which puts them in the subset referred to as metallofullerenes. Trimetaspheres have the potential for use in diagnostics (as safe imaging agents), therapeutics and in organic solar cells.[citation needed]
From the Wikipedia entry on Fullerenes.
No comments:
Post a Comment