A related term is thermal energy, loosely defined as the energy of a body that increases with its temperature. Heat is also loosely referred to as thermal energy, although many definitions require this thermal energy to actually be in the process of movement between one body and another to be technically called heat (otherwise, many sources prefer to continue to refer to the static quantity as "thermal energy"). Heat is also known as "Energy".
Energy transfer by heat can occur between objects by radiation, conduction and convection. Temperature is used as a measure of the internal energy or enthalpy, that is the level of elementary motion giving rise to heat transfer. Energy can only be transferred by heat between objects - or areas within an object - with different temperatures (as given by the zeroth law of thermodynamics). This transfer happens spontaneously only in the direction of the colder body (as per the second law of thermodynamics). The transfer of energy by heat from one object to another object with an equal or higher temperature can happen only with the aid of a heat pump via mechanical work.
Contents |
Overview
The first law of thermodynamics states that the energy of an isolated system is conserved. Therefore, to change the energy of a system, energy must be transferred to or from the system. Heat and work are the only two mechanisms by which energy can be transferred to or from a control mass. Work performed on a body is, by definition [1] an energy transfer to the body that is due to a change of the external parameters of the body (such as the volume, magnetization, center of mass position in a gravitational field etc.). Heat is the energy transferred to the body in any other way. This definition of heat applies generally: it does not appeal to any notion of thermal equilibrium.In case of bodies close to thermal equilibrium where notions such as the temperature can be defined, heat transfer can be related to temperature difference between bodies. It is an irreversible process that leads to the bodies coming closer to mutual thermal equilibrium.
The unit for the amount of energy transferred by heat in the International System of Units SI is the joule (J), though the British Thermal Unit and the calorie are still used in the United States. The unit for the rate of heat transfer is the watt (W = J/s).
Heat transfer is a path function (process quantity), as opposed to a point function (state quantity). Heat flows between systems that are not in thermal equilibrium with each other; it spontaneously flows from the areas of high temperature to areas of low temperature. When two bodies of different temperature come into thermal contact, they will exchange internal energy until their temperatures are equalized; that is, until they reach thermal equilibrium. The adjective hot is used as a relative term to compare the object’s temperature to that of the surroundings (or that of the person using the term). The term heat is used to describe the flow of energy. In the absence of work interactions, the heat that is transferred to an object ends up getting stored in the object in the form of internal energy.

A red-hot iron rod from which heat transfer to the surrounding environment will be primarily through radiation.
Notation
The total amount of energy transferred through heat transfer is conventionally abbreviated as Q. The conventional sign convention is that when a body releases heat into its surroundings, Q < 0 (-); when a body absorbs heat from its surroundings, Q > 0 (+). Heat transfer rate, or heat flow per unit time, is denoted by: .
.
Entropy
In 1854, German physicist Rudolf Clausius defined the second fundamental theorem (the second law of thermodynamics) in the mechanical theory of heat (thermodynamics): "if two transformations which, without necessitating any other permanent change, can mutually replace one another, be called equivalent, then the generations of the quantity of heat Q from work at the temperature T, has the equivalence-value:"[2][3]Definitions
In modern terms, heat is concisely defined as energy in transit. Scottish physicist James Clerk Maxwell, in his 1871 classic Theory of Heat, was one of the first to enunciate a modern definition of “heat”. In short, Maxwell outlined four stipulations on the definition of heat. One, it is “something which may be transferred from one body to another”, as per the second law of thermodynamics. Two, it can be spoken of as a “measurable quantity”, and thus treated mathematically like other measurable quantities. Three, it “can not be treated as a substance”; for it may be transformed into something which is not a substance, e.g. mechanical work. Lastly, it is “one of the forms of energy”. Similar such modern, succinct definitions of heat are as follows:- In a thermodynamic sense, heat is never regarded as being stored within a body. Like work, it exists only as energy in transit from one body to another; in thermodynamic terminology, between a system and its surroundings. When energy in the form of heat is added to a system, it is stored not as heat, but as kinetic and potential energy of the atoms and molecules making up the system.[4]
- The noun heat is defined only during the process of energy transfer by conduction or radiation.[5]
- Heat is defined as any spontaneous flow of energy from one object to another, caused by a difference in temperature between the objects.[6]
- Heat may be defined as energy in transit from a high-temperature object to a lower-temperature object.[7]
- Heat as an interaction between two closed systems without exchange of work is a pure heat interaction when the two systems, initially isolated and in a stable equilibrium, are placed in contact. The energy exchanged between the two systems is then called heat.[8]
- Heat is a form of energy possessed by a substance by virtue of the vibrational movement, i.e. kinetic energy, of its molecules or atoms.[9] The kinetic energy and heat may formally be equivalent, but they are not identical.
- Heat is the transfer of energy between substances of different temperatures.
Thermodynamics
Internal energy
Heat is related to the internal energy U of the system and work W done by the system by the first law of thermodynamics:| Type | Composition of Internal Energy (U) |
|---|---|
| Sensible energy | the portion of the internal energy of a system associated with kinetic energies (molecular translation, rotation, and vibration; electron translation and spin; and nuclear spin) of the molecules. |
| Latent energy | the internal energy associated with the phase of a system. |
| Chemical energy | the internal energy associated with the atomic bonds in a molecule. |
| Nuclear energy | the tremendous amount of energy associated with the strong bonds within the nucleus of the atom itself. |
| Energy interactions | those types of energies not stored in the system (e.g. heat transfer, mass transfer, and work), but which are recognized at the system boundary as they cross it, which represent gains or losses by a system during a process. |
| Thermal energy | the sum of sensible and latent forms of internal energy. |
Heat capacity
For a simple compressible system such as an ideal gas inside a piston, the changes in enthalpy and internal energy can be related to the heat capacity at constant pressure and volume, respectively. Constrained to have constant volume, the heat, Q, required to change its temperature from an initial temperature, T0, to a final temperature, Tf is given by: according to:
according to: according to:
according to:The specific heats of monatomic gases (e.g., helium) are nearly constant with temperature. Diatomic gases such as hydrogen display some temperature dependence, and triatomic gases (e.g., carbon dioxide) still more.
In liquids at sufficiently low temperatures, quantum effects become significant. An example is the behavior of bosons such as helium-4. For such substances, the behavior of heat capacity with temperature is discontinuous at the Bose-Einstein condensation point.
The quantum behavior of solids is adequately characterized by the Debye model. At temperatures well below the characteristic Debye temperature of a solid lattice, its specific heat will be proportional to the cube of absolute temperature. For low-temperature metals, a second term is needed to account for the behavior of the conduction electrons, an example of Fermi-Dirac statistics.
Phase Changes
The boiling point of water, at sea level and normal atmospheric pressure and temperature, will always be at nearly 100 °C, no matter how much heat is added. The extra heat changes the phase of the water from liquid into water vapor. The heat added to change the phase of a substance in this way is said to be "hidden" and thus it is called latent heat (from the Latin latere meaning "to lie hidden"). Latent heat is the heat per unit mass necessary to change the state of a given substance, or:- Q = LΔm.
Heat transfer mechanisms
Main article: Heat transfer
Heat tends to move from a high-temperature region to a low-temperature region. This heat transfer may occur by the mechanisms of conduction and radiation. In engineering, the term convective heat transfer is used to describe the combined effects of conduction and fluid flow and is regarded as a third mechanism of heat transfer.Conduction
Conduction is the most significant means of heat transfer in a solid. On a microscopic scale, conduction occurs as hot, rapidly moving or vibrating atoms and molecules interact with neighboring atoms and molecules, transferring some of their energy (heat) to these neighboring atoms. In insulators the heat flux is carried almost entirely by phonon vibrations.
Fire test used to test the heat transfer through firestops and penetrants used in construction listing and approval use and compliance.
Convection
Convection is usually the dominant form of heat transfer in liquids and gases. This is a term used to characterise the combined effects of conduction and fluid flow. In convection, enthalpy transfer occurs by the movement of hot or cold portions of the fluid together with heat transfer by conduction. Commonly an increase in temperature produces a reduction in density. Hence, when water is heated on a stove, hot water from the bottom of the pan rises, displacing the colder denser liquid which falls. Mixing and conduction result eventually in a nearly homogeneous density and even temperature. Two types of convection are commonly distinguished, free convection, in which gravity and buoyancy forces drive the fluid movement, and forced convection, where a fan, stirrer, or other means is used to move the fluid. Buoyant convection is due to the effects of gravity, and hence does not occur in microgravity environments.Radiation
Radiation is the only form of heat transfer that can occur in the absence of any form of medium (i.e., through a vacuum). Thermal radiation is a direct result of the movements of atoms and molecules in a material. Since these atoms and molecules are composed of charged particles (protons and electrons), their movements result in the emission of electromagnetic radiation, which carries energy away from the surface. At the same time, the surface is constantly bombarded by radiation from the surroundings, resulting in the transfer of energy to the surface. Since the amount of emitted radiation increases with increasing temperature, a net transfer of energy from higher temperatures to lower temperatures results.The power that a black body emits at various frequencies is described by Planck's law. For any given temperature, there is a frequency fmax at which the power emitted is a maximum. Wien's displacement law, and the fact that the frequency of light is inversely proportional to its wavelength in vacuum, mean that the peak frequency fmax is proportional to the absolute temperature T of the black body. The photosphere of the Sun, at a temperature of approximately 6000 K, emits radiation principally in the visible portion of the spectrum. The Earth's atmosphere is partly transparent to visible light, and the light reaching the Earth's surface is absorbed or reflected. The Earth's surface emits the absorbed radiation, approximating the behavior of a black body at 300 K with spectral peak at fmax. At these lower frequencies, the atmosphere is largely opaque and radiation from the Earth's surface is absorbed or scattered by the atmosphere. Though some radiation escapes into space, it is absorbed and subsequently re-emitted by atmospheric gases. It is this spectral selectivity of the atmosphere that is responsible for the planetary greenhouse effect.
The common household lightbulb has a spectrum overlapping the blackbody spectra of the sun and the earth. A portion of the photons emitted by a tungsten light bulb filament at 3000K are in the visible spectrum. However, most of the energy is associated with photons of longer wavelengths; these will not help a person see, but will still transfer heat to the environment, as can be deduced empirically by observing a household incandescent lightbulb. Whenever EM radiation is emitted and then absorbed, heat is transferred. This principle is used in microwave ovens, laser cutting, and RF hair removal.
Other heat transfer mechanisms
- Latent heat: Transfer of heat through a physical change in the medium such as water-to-ice or water-to-steam or steam-to-water or ice-to-water involves significant energy and is exploited in many ways: steam engine, refrigerator etc. (see latent heat of fusion)
- Heat pipes: Using latent heat and capillary action to move heat, heat pipes can carry many times as much heat as a similar-sized copper rod. Originally invented for use in satellites, they are starting to have applications in personal computers.
Heat dissipation
In cold climates, houses with their heating systems form dissipative systems. In spite of efforts to insulate such houses to reduce heat losses to their exteriors, considerable heat is lost, or dissipated, from them, which can make their interiors uncomfortably cool or cold. For the comfort of its inhabitants, the interior of a house must be maintained out of thermal equilibrium with its external surroundings. In effect, domestic residences are oases of warmth in a sea of cold and the thermal gradient between the inside and outside is often quite steep. This can lead to problems such as condensation and uncomfortable draughts (drafts) which, if left unaddressed, can cause structural damage to the property. This is why modern insulation techniques are required to reduce heat loss.In such a house, a thermostat is a device capable of starting the heating system when the house's interior falls below a set temperature, and of stopping that same system when another (higher) set temperature has been achieved. Thus the thermostat controls the flow of energy into the house, that energy eventually being dissipated to the exterior.
References
- ^ a b F. Reif (2000). Fundamentals of Statistical and Thermal Physics. Singapore: McGraw-Hll, Inc.. p. 66. ISBN 0-07-Y85615-X.
- ^ Published in Poggendoff’s Annalen, Dec. 1854, vol. xciii. p. 481; translated in the Journal de Mathematiques, vol. xx. Paris, 1855, and in the Philosophical Magazine, August 1856, s. 4. vol. xii, p. 81
- ^ Clausius, R. (1865). The Mechanical Theory of Heat] – with its Applications to the Steam Engine and to Physical Properties of Bodies. London: John van Voorst, 1 Paternoster Row. MDCCCLXVII.
- ^ Smith, J.M., Van Ness, H.C., Abbot, M.M. (2005). Introduction to Chemical Engineering Thermodynamics. McGraw-Hill. ISBN 0073104450.
- ^ Baierlein, Ralph (2003). Thermal Physics. Cambridge University Press. ISBN 0521658381.
- ^ Schroeder, Daniel V. (2000). An introduction to thermal physics. San Francisco, California: Addison-Wesley. p. 18. ISBN 0-321-27779-1. "Heat is defined as any spontaneous flow of energy from one object to another, caused by a difference in temperature between the objects."
- ^ Discourse on Heat and Work - Department of Physics and Astronomy, Georgia State University: Hyperphysics (online)
- ^ Perrot, Pierre (1998). A to Z of Thermodynamics. Oxford University Press. ISBN 0198565526.
- ^ Clark, John, O.E. (2004). The Essential Dictionary of Science. Barnes & Noble Books. ISBN 0760746168.
- ^ Cengel, Yungus, A.; Boles, Michael (2002). Thermodynamics: An Engineering Approach (4th ed.). Boston: McGraw-Hill. pp. 17–18. ISBN 0-07-238332-1.
See also
External links
- Plasma heat at 2 gigakelvins - Article about extremely high temperature generated by scientists (Foxnews.com)
- Correlations for Convective Heat Transfer - ChE Online Resources
- An Introduction to the Quantitative Definition and Analysis of Heat written for High School Students

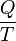
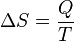


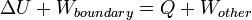
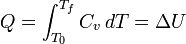
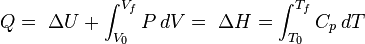


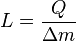
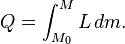
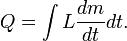

No comments:
Post a Comment